Adopting New Strategies for Specialty Savings
It seems that every other month there’s a new specialty medication, one that’s high-cost and requires a complex distribution strategy, hitting the market. They make formulary planning difficult and disrupt long-term budgeting. And until 2015, they were the only choice for patients needing these medications.
So what has changed in the years since then?
Biosimilars entered the market.
A Change in Price, Not Quality
Biosimilars are the U.S. Food and Drug Administration (FDA)-approved alternatives to the originator (or reference) products. They match brand name drugs in safety, efficacy and quality, and their primary purpose is the same as ours: to drive affordability without compromising care.
One of the barriers these drugs are running into is adoption rates. While the rates are rising steadily, it remains uneven across different classes of drugs, mainly due to these three reasons:
- Provider hesitancy – Concerns about interchangeability and clinical outcomes can slow prescribing rates
- Lack of patient education – There are many misconceptions about safety and efficacy, leading to hesitancy to change medications
- Contracting challenges – Complex rebate structures can disincentivize biosimilar usage, meaning non-pass-through PBMs would have to forego their spread pricing
Even with these barriers in place, at least one category, targeted immunomodulators (TIMs), has experienced multiple years of declining net costs thanks mostly to biosimilars.1 They not only offer more budget-friendly alternatives to name brand TIMs, but by being in the market, they force the originator products to lower their own prices to stay competitive.
Another ongoing issue is regulatory approvals. While the FDA has approved dozens of biosimilars, and has many more in the pipeline, it takes a long time to get one to market. In fact, it takes approximately 7-9 years and carries a cost in the hundreds of millions.2 But there is hope on the horizon. The FDA is trying to accelerate the development of biosimilars through a streamlined process. Through these adjustments, we could see drug development time cut in half.
Every Biosimilar is a Cure for High Prices
There are projections that the global biosimilar market will exceed $100 billion by 2030, which means we could see the overall prices come down as reference drugs try to stay ahead of their biosimilars. While there’s no exact figure for how much a plan sponsor can save by adopting biosimilars over their reference drugs, it usually starts at 15% and goes up to 50%. When Navitus added Humira® biosimilars to our formularies in 2023, then took the added step of removing Humira in favor of biosimilars, we were able to reduce net cost claims by an astounding 60%.1 And when we removed Stelara® from our formulary in 2025, we were able to offer plan sponsors tens of thousands of dollars in net savings per dose.
That means reduced costs for plan sponsors and more affordable medications for members. Unlike traditional PBMs, who merely moved the brand name drugs to a non-preferred tier, Navitus takes pride in maximizing these biosimilar savings by completely removing the brand name drugs from formulary, leading to much higher adoption rates. These widespread adoptions could easily translate into billions of dollars in annual savings.
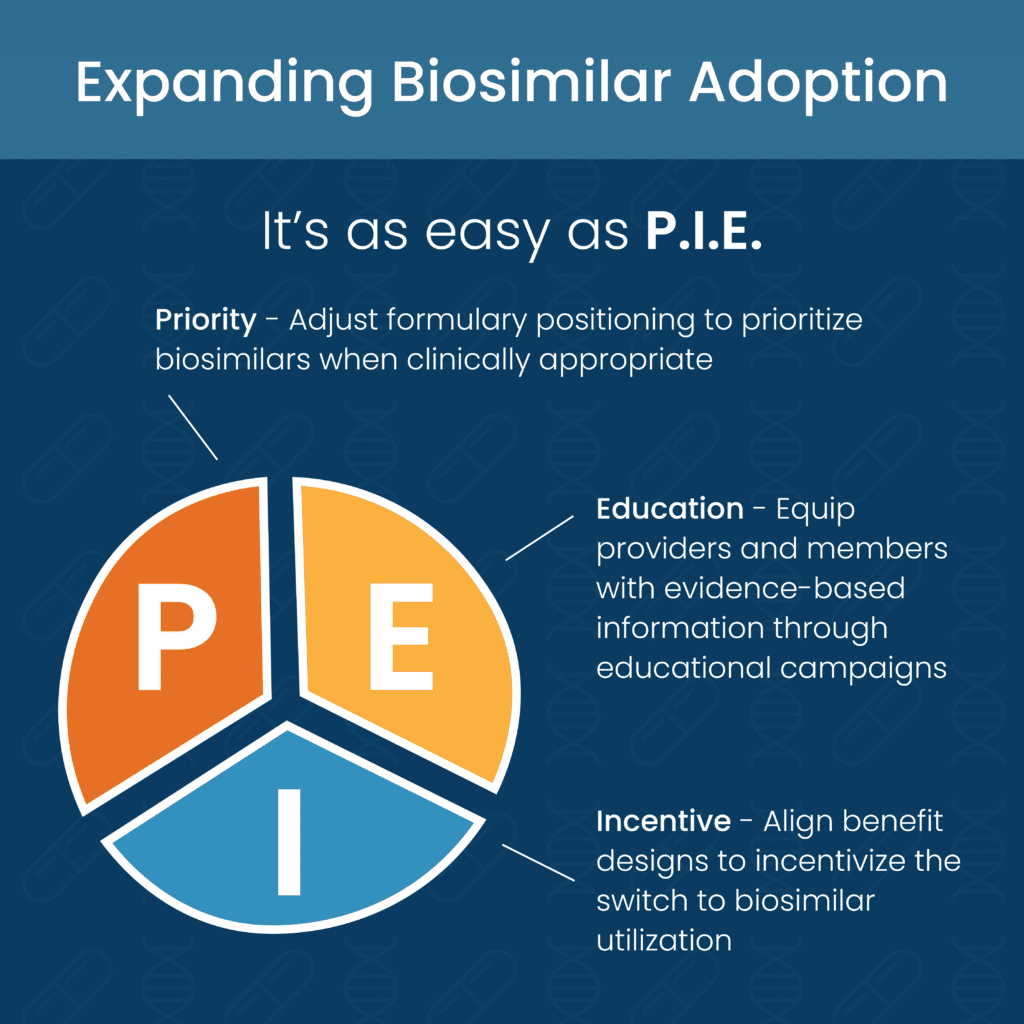
But the expanded adoption of biosimilars is not all dependent upon PBMs. It’s also up to plan sponsors to take charge of their future, and we’ve made is as easy as P.I.E.:
- Priority – Adjust formulary positioning to prioritize biosimilars when clinically appropriate
- Incentive – Align benefit decisions to incentivize the switch to biosimilar utilization
- Education – Equip providers and members with evidence-based information through educational campaigns
Start A Conversation
Ready to explore how biosimilars can reduce costs without sacrificing quality of care? Reach out to [email protected] to get started.
References
1. 2024 Navitus Drug Trend Report. www.navitus.com/drug-trend-reports/2024-snapshot/
2. Tuszyner A, The New Era of Biosimilar Development: Seizing the Opportunity Under EMA’s Streamlined Guidelines. The New Era of Biosimilar Development | Science Hub | Mabion. Updated 2025. Accessed January 22, 2026.
Stay Informed and Connected
Receive expert insights, healthcare tips, and important updates on pharmacy benefits, drug recalls, and more—straight to your inbox.
CLARITY FOR ACTION
Gain Clarity: Navitus 10th Annual Drug Trend Report Ready to Download
Our 2025 report provides transparent, data-driven insight to help plans, plan sponsors and consultants navigate an increasingly complex prescription drug landscape.